Abstract
Introduction
Rozanolixizumab is a humanized, high-affinity, anti-human neonatal Fc receptor (FcRn) monoclonal antibody, developed with the aim of reducing levels of pathogenic immunoglobulin G (IgG) in autoimmune and alloimmune diseases. In a first-in-human (FIH) healthy subject trial (NCT02220153), single-dose rozanolixizumab was well tolerated at doses up to 4 mg/kg intravenous (IV) and 7 mg/kg subcutaneous (SC) (Kiessling P et al. P3_48, Peripheral Nerve Society Annual Meeting, Sitges, Spain, 11 July 2017). Severe treatment-emergent adverse events (TEAEs; headache and back pain) occurred with 7mg/kg IV. No severe TEAEs were reported with SC administration. IgG concentrations were reduced by up to 48%, with the greatest mean reduction at maximum dose reached by Day 10, returning to baseline by Day 57.
Here we report an interim analysis (5th Data Monitoring Committee review) of an ongoing Phase II, open-label study evaluating the safety, tolerability and efficacy of rozanolixizumab in patients (pts) with primary persistent or chronic immune thrombocytopenia (ITP) (NCT02718716).
Methods
Eligibility criteria included: ≥18 years of age, diagnosis of primary ITP for a minimum of 3 months prior to screening, platelet count <30x109/L at screening and <35x109/L at baseline, current or history of a peripheral blood smear consistent with ITP. Eligible pts received weekly doses of SC rozanolixizumab (5x4 mg/kg), following a review of pt responses, the weekly dose was escalated (3x7 mg/kg). The primary objective is to evaluate the safety and tolerability of rozanolixizumab. Interim analysis data cut-off: 21 February 2017.
Results
A total of 28 pts received rozanolixizumab: 4 mg/kg (n=15), 7 mg/kg (n=13). Median age was 56.0 years (range 20-86); 4 mg/kg 66.0 (21-86) and 7 mg/kg 54.0 (20-73). Median duration of disease at baseline was 5.6 years; 4 mg/kg 7.1 and 7 mg/kg 3.8. Of 26 pts who received prior ITP therapies, median number of therapies was 4.0 (range 1-15); 4 mg/kg 4.0 (1-15; n=14) and 7 mg/kg 3.5 (1-12; n=12). Most common therapies (by preferred term): azathioprine (12/28 patients, 42.9%; 4 mg/kg 46.7% and 7 mg/kg 38.5%), romiplostim (11/28, 39.3%; 4 mg/kg 46.7% and 7 mg/kg 30.8%), immunoglobulins (10/28, 35.7%; 4 mg/kg 33.3% and 7 mg/kg 38.5%).
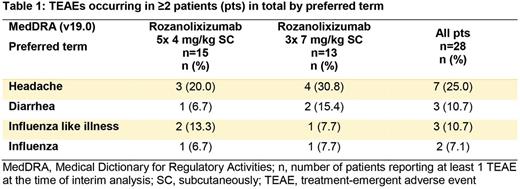
Overall, 18/28 (64.3%) pts reported at least 1 TEAE (combined total of 55 TEAEs); 12/15 (80.0%) pts in the 4 mg/kg group and 6/13 (46.2%) pts in the 7 mg/kg group. Apart from 1 severe (Common Terminology Criteria for Adverse Events [CTCAE] Grade 3 or above) TEAE (bleeding from genital tract) in the 4 mg/kg group, deemed unrelated to study medication by the investigator, all TEAEs reported were mild/moderate (CTCAE Grade 1/2). One pt reported a TEAE deemed related to study medication by the investigator: CTCAE Grade 1 injection site reaction in the 4 mg/kg group, which did not interfere with subsequent dosing. The most frequently reported TEAE was headache (CTCAE Grade 1): 3/15 (20.0%) pts in 4 mg/kg group and 4/13 (30.8%) pts in 7 mg/kg group (Table 1). Anti-drug antibodies were detected in 19/28 pts with no effects on rozanolixizumab pharmacodynamics; 3 cases considered treatment emergent. No clinically relevant changes were observed in all other hematology, coagulation, clinical chemistry (including albumin), urinalysis, ECGs, vital signs or liver function tests. No opportunistic infections were reported. No treatment discontinuations due to TEAEs or TEAEs leading to death were reported.
At the interim analysis, maximum mean decreases in total IgG levels were observed at Day 29 for the 4 mg/kg group (43.6%, range 21.9-68.6) and at Day 22 for the 7 mg/kg group (50.5%, 35.7-65.5). Clinically relevant improvement in platelet counts (values ≥50x109/L) were reported for 8 pts (53.3%) in the 4 mg/kg group (maximum value range: 50x109/L to 198x109/L) and 4 pts (30.8%) in the 7 mg/kg group (59x109/L to 133x109/L). In this population of responders, maximal reductions in IgG levels were observed, ranging from 23.9-68.6% in the 4 mg/kg group and 51.1-65.5% in the 7 mg/kg group.
Conclusion
From the data available to date, multiple dosing with rozanolixizumab 4 mg/kg SC and 7 mg/kg SC has been well tolerated in pts with ITP, and initial platelet responses have been observed in both dose groups. The safety profile for SC rozanolixizumab reported in this trial is in line with the safety profile reported in the FIH trial with healthy subjects receiving SC rozanolixizumab.
Robak: UCB Pharma: Research Funding. Musteata: Artensia EM: Other: Principal investigator of the multicenter, open-label, multiple-dose clinical study. Kiessling: UCB Pharma: Employment. Massow: UCB Pharma: Employment. Higginson: UCB Pharma: Employment, Equity Ownership. Snipes: UCB Pharma: Employment. Jolles: SOBI & Binding Site: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Drug Safety Monitoring Committee, Immunodeficiency Teaching Academy, Research Funding, Speakers Bureau; LFB: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Drug Safety Monitoring Committee, Immunodeficiency Teaching Academy, Research Funding, Speakers Bureau; Biotest: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Drug Safety Monitoring Committee, Immunodeficiency Teaching Academy, Research Funding, Speakers Bureau; BPL: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Drug Safety Monitoring Committee, Immunodeficiency Teaching Academy, Research Funding, Speakers Bureau; Grifols: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Drug Safety Monitoring Committee, Immunodeficiency Teaching Academy, Research Funding, Speakers Bureau; Octapharma: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Drug Safety Monitoring Committee, Immunodeficiency Teaching Academy, Research Funding, Speakers Bureau; CSL Behring: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Drug Safety Monitoring Committee, Immunodeficiency Teaching Academy, Research Funding, Speakers Bureau; Shire: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Drug Safety Monitoring Committee, Immunodeficiency Teaching Academy, Research Funding, Speakers Bureau; UCB Pharma: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Drug Safety Monitoring Committee, Immunodeficiency Teaching Academy, Research Funding, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal